What the curriculum thinks you need to know:
PC_BK_17 Humidity, absolute and relative; including measurement
PC_BK_18 Colligative properties: osmolarity, osmolality, osmometry, diffusion
What you need to know (The theory):
This is all about dissolving stuff into other stuff… Its great exam fodder as there is…
Definitions, Definitions, LOTS of Definitions….
Humidity
The amount of water (vapour) present within a gas (usually air).
Absolute Humidity
The mass of water vapour (Grams) present per volume of air (Cubic Metre).
Relative Humidity
Ratio of water vapour present against the maximum possible (e.g. fully saturated)
Relative humidity is usually expressed as a percentage and is what you tend to hear in a weather report.
Relative humidity is:
RH = (Actual Vapour Pressure) / (Saturation Vapour Pressure) X 100%
Its important to remember that the amount of water vapour a volume of gas is dependant on a number of factors. These are exactly the same factors that determine how much anaesthetic vapour can be held by a volume of gas…
Namely….
Ambient Pressure – The higher the ambient pressure, the less water can be held.
Ambient Temperature – The higher the temperature, the more water can be held.
This explains why you get early morning Dew forming when the temperature drops as the air can no longer ‘hold’ the water, so it condenses out.
Dew point
Temperature below which water will begin to condense out of air. (e.g. relative humidity reaches 100%)
How do we measure it though?
People with long hair will probably notice your hair gets longer when it wet. This principle was used in the hair hygrometer. A piece of hair is secured at one end, and then has a small weight at the bottom end. This is then placed in the place you wish to know the humidity. The higher the humidity the longer the hair.
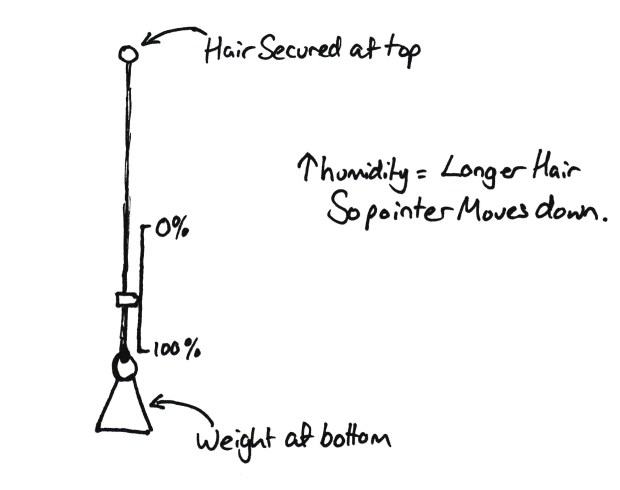
Simple hair hygrometer
Having bits of hair hanging around the place is hardly elegant, so people began coming up with other solutions. You’ll probably notice that clothes on a washing line dry quicker when its warmer (obviously) but also when where they are drying is less humid. This is because the surrounding air has ‘room’ for water to evaporate. If you take this to the extreme, Clothes will never dry in a rainforest (close to 100% humidity), but will, eventually, dry in a freezer (-20oC with low humidity). So we know that water evaporates from something wet when ambient humidity is less….
The wet and dry hygrometer uses this principle. It uses two thermometers… One has its bulb open to air, the other is wrapped in a wet rag. Now we know from this article that for a substance to vapourise/evaporate we need energy. This usually comes from cooling its surroundings (e.g. stealing heat energy). This is the same reason why your sevoflurane vapouriser is freezing when you have it at 8% with a high fresh gas flow. So if we are in a area of low humidity, out thermometer with a wet rag around it is going to cool down as water is evaporated into the air. The closer the relative humidity is to 100%, the less water evaporates so the closer the temperature is to the thermometer without a wet rag. Remember, the closer the temperature between the two thermometers, the higher the relative humidity. At 100% relative humidity, there should be no difference between the readings as no water will evaporate.

Wet/Dry Hygrometer
Note you may see a wet/dry hygrometer referred to as a psychrometer.
Modern Hygrometers tend to use either capacitive or resistive methods to measure relative humidity. These work by knowing how the physical properties of a semiconductor change with humidity. They typically need calibration and are subject to things like drift.
An important way of measuring absolute humidity is using an optical method looking at transmission of light through a sample. This is VERY similar to how your infrared spectrograph works to give a capnography trace (see here). But typically these tend to use lasers rather than infrared light. But, the principle is identical otherwise.
Colligative Properties.

Colligative properties – We all use them.
Think about a cold winters day… The salt spreaders are out. But why?
Salt disolved into water lowers its freezing point. So less ice, less slipping over and less car accidents.
When we dissolve a solute (e.g. salt) into a solvent (e.g. water) it has a number of measurable effects. Its worth noting these effects are dependant on the number of solute molcules, not their type.
- decreases the solutes freezing point (as we’ve already said)
- Increases the solutes boiling point (the reason you put salt in your water when cooking pasta, it increases the boiling point, makes the water hotter and cooks your pasta faster)
- Decreases the SVP which the solvent can attain (this stands to reason as there is less space for solvent molecules on the surface of the solvent, so less can vapourise easily).
- The solute exerts an osmotic effect.
Now for more definitions:
Mole
‘a cute furry creature that lives in a hole’ or ‘6.022×10^23 molecules of a substance’
Molarity
Number of molecules of solute per litre of solution
Molality
Number of molecules of solute per kilogram of solvent
Osmole
6.022×10^23 osmotically active particles
Note: one mole of NaCl will equal two Osmoles as the NaCl will dissociate in solution giving twice as many osmotically active particles.
Osmolarity (one L!)
Number of osmoles of solute per litre of solution
Osmolality (two L’s!)
Number of osmoles of solute per kilogram of solvent
Great, But why do we care?
Solutions which have a high osmolarity/osmolality will attract water. This is used in many biological systems (e.g. the kidney). The opposite is also true, a solution with a low osmolality will attract solute. This is the principle behind osmosis.
Diffusion
Movement of a substance/solute from an area of high concentration to that of a low concentration
Osmosis
Movement of a solvent across a semipermeable membrane from a less concentrated solution to a more concentrated solution.
What you need to know (How it works in practice):
Although we rarely actively measure humidity in practice, its useful to know about it. Why? Mostly around the respiratory system. Our nose is one of the greatest humidifiers known to man. air we breathe in is almost totally humidified before it hits our lower airways. This helps stop damage to the mucosal layers and maintain their function. So what do we do as anaesthetists? bypass it. So we need to know how to humidify the air we deliver to patients.
Methods of humidification:
HME – Hydroscopic material ‘captures’ moisture on exhalation. Then it is ‘picked’ up again on inspiration. Not totally effective, but mostly humidifies fresh gas flow after a period of ‘warm up’.
Nebulisers/bubble humifier – Bubble gas through water or vapourise water with a pressurised gas flow.
Heated Humidifier – Think of these (e.g. fisher paykel humidifier) as a sevoflurane vapouriser for water. Water is heated in a dish, and fresh gas flow passes over it so it rapidly humidifies and heats the gas flow.
Random Exam factoids (i.e. the things the college like asking):
- The main things here are the definitions. They make excellent MCQs. Learn them well. The small differences between the definitions need to be remembered!
© Sam Beckett and Physics4FRCA, 2018. Unauthorized use and/or duplication of this material without express and written permission from this site’s author and/or owner is strictly prohibited.
