What the curriculum thinks you need to know:
Demonstrates knowledge of:
PC_BK_71 Capnography
PC_BK_78 Measurement of gas and vapour concentrations: e.g. infra-red,paramagnetic, fuel cell, oxygen electrode, mass spectrometry
This post will concentrate on *most* of the gas monitoring you will actually use in the anaesthetic room/theatre, not the inner workings of a gas machine or lab based systems.
What you need to know (the theory):
CO2, N20 and Halogenated agent monitoring (e.g everything apart from oxygen) operates around the absorption spectroscopy principle, specifically, absorption of infrared light:
Any molecule made up of one or more dissimilar atoms (polyatomic) will absorb infrared light in a given wavelength range.
So if we first look at CO2, it has a wide absorption spectra, but its peak absorption is at 4.28μm. Absoprtion at other wavelengths is low. So we use this peak wavelength as its easier to measure due to the large absorption of light.
If we take a sample of gas of known size and shine light through it (usually generated by a hot wire), some of that light will be absorbed by the gas molecules. The amount of light which passes through can then be measured using a photo detector. The amount of light detected in inversely proportional to the concentration of gas present (as more light is absorbed if more gas molecules are present)
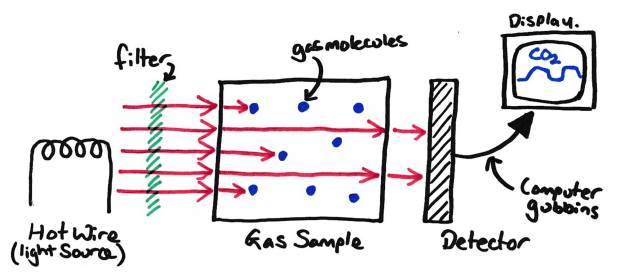
A Basic Infrared Gas Analyser
If we then filter the incoming light to a specific wavelength with a filter (in this case only letting light with a wavelength of around 4.28μm through), we can restrict the light available to that which is absorbed by CO2. This allows us to measure the amount of CO2 present in a gas mixture, as the other gases won’t absorb (much) light at that specific wavelength.
The amount of light absorbed by the gas is dependant on the number of molecules the light will come in contact with, and will be proportional to a few things:
Lambert’s Law
The absorption of light is proportional to the distance the light travels through the sample
To remember this one, just think of someone smoking a cigarette (A Lambert and Butler for instance!), The more ash on the end of the cigarette, the less you can see the burning tobacco glow, as the light has to travel through more ash to be seen.
Beer’s Law
The absorption of light is proportional to the concentration of the gas through which the light is travelling
To remember this one, just think of beer. Stronger beer (generally) is darker in colour, so the higher the concentration of alcohol the less the light gets through (No, ethanol doesn’t cause the colour in beer, but its a way to remember it!)
Gas Pressure
The absorption of light is proportional to the pressure of the gas sample (the higher the pressure, the more gas molecules in a certain volume so the higher the absorption)
As we know the distance travelled by the light is constant (the sample chamber size doesn’t change), as is the sample pressure, so the absorption of light is proportional only to the concentration of gas present. If we hook up the photo detector on the other side of the sample to a computer and we plot the voltage produced from this we get a graph which is proportional to the concentration of the gas present in the sample chamber. This is the typical capnograph we see day-to-day.
What you need to know (In Practice):
So we’ve said we can measure CO2 with this setup, but how do we measure the other gases? Simple, we change the wavelength of the emitted light by changing the wavelength the filter lets through to one specific for another agent (e.g. isoflurane, desflurane etc). This is either done by having separate light emitters/filter/detector sets for each agent we are interested in, OR by having a rotating window which changes the filter multiple times a second. This allows monitoring of multiple agents at the same time.
We’ve shown how we analyse the gas content in a sample, but how do we monitor the gas going into and out of the patient? The monitor can be in one of two places in the anaesthetic circuit:
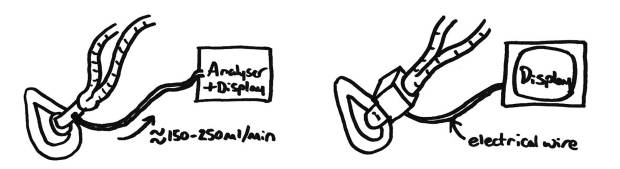
Sidestream (Left) vs Mainstream (Right) Gas Analysers
Sidestream analyser –
- Takes a small sample tube from the patient end of the circuit (typically attached to the HME filter) and withdraws 150-250ml/min of gas from here to analyse. This can then be added back to the circle system after analysis to aid in the usage of low flows and minimise the amount of volatiles used.
- Allows the monitoring equipment to be moved off the patient, to the anaesthetic machine.
- Allows larger analysers with more agents measured.
- The length of tube is proportional to the delay seen between a patient breath and the response on the capnograph. Regardless, this is a much larger delay than in mainstream analysers. The delay can be minimised by using a higher draw rate of gas from the circle, shorter sample tubing and smaller diameter tubing (this will then hold less gas).
Mainstream analyser –
- Attached directly to the breathing circuit, so little to no delay in response.
- Clips around a clear plastic window in the breathing circuit.
- Usually small and portable, so great for transferring patients or out of hospital situations.
- Typically only measure CO2 due to their small size and the fact they are usually reliant on battery power.
- Can get hot, so take care with proximity to patient.
- Can be unreliable due to water vapour problems (see below).
Regardless of position, both need to have their sample source (be this the sample port on a side stream analyser or the window for a mainstream analyser) as close to the patient as possible. This minimised dead space and ensures that the response of the capnograph is accurate.
Problems with absorption spectroscopy:
- Water – Water can cause a physical blockage of sample lines, this is mostly seen in ITU situations where a HME is not used in a humidified circuit. This can be reduced by pointing the sample tube so it exits the circuit upwards, but this still doesn’t eliminate problems. Can also cause problems if it accumulates on the window in a mainstream analyser.
- Water Vapour – Water vapour can cause inaccurate readings as it will absorb infrared light across a wide spectrum. To try to stop this, a water trap is usually present before the analyser in a side stream setup. Obviously this is not possible in a main stream setup.
- Nitrous oxide – The analyser can be used to measure nitrous concentrations, however, it does cause a problem with CO2 measurement due to the ‘collision broadening effect’ (There are a few explanations of this, but the following is the simplest). This is where nitrous oxide collides with CO2 molecules which have already absorbed some light energy and ‘takes’ the energy off them. This then allows the CO2 to absorb more light energy giving a falsely high reading for CO2 concentration. Modern anaesthetic machines have built-in compensation for this collision broadening effect, predicting how much it will occur given a particular nitrous concentration.
Typical capnograph traces
** CO2 traces **
Causes of Low etCO2:
- Hyperventilation (anxiety, too fast ventilation rate on ventilator etc)
- Low cardiac output
- Leaking circuit
- Cardiac Arrest or similar (gradual drop of CO2 over a few breaths)
- Disconnection from circuit (instantaneous loss of CO2 trace)
- Gas monitor zeroing (the most common cause!!)
Causes of High etCO2
- Sepsis
- Malignant Hyperpyrexia
- Endocrine causes (thyroid storm etc)
- Exogenous CO2 (think laproscopic surgery)
Random Exam factoids (i.e. the things the college like asking):
- The sample analyser is lined in sapphire, as this does not absorb infrared light, whilst glass would.
- Carbon dioxide has a peak absorption spectra of 4.28μm
© Sam Beckett and Physics4FRCA, 2017. Unauthorized use and/or duplication of this material without express and written permission from this site’s author and/or owner is strictly prohibited.