What the curriculum thinks you need to know:
PC_BK_72 Pulse oximetry
Vague Huh!?!
What you need to know (The theory):
If you’ve read the post on infrared absorption spectroscopy, then you already know most of this. I recommend you read this post first.
We learnt that any atom with two or more dissimilar atoms (polyatomic) will absorb infrared light at a given wavelength.
Pulse oximeters work along the same principles as gas analysers, but are slightly more complicated. The concept of infrared absorption works for both gas samples and solid samples. Just hold your hand upto the sun, it blocks most infra red!
If we take a sample of blood in a bottle, the Oxyhaemoglobin and deoxyhaemoglobin in the sample will absorb infrared light along the same wavelength range, but they each have different peak absorption wavelengths. They also have a wavelength at which their absorption is the same.
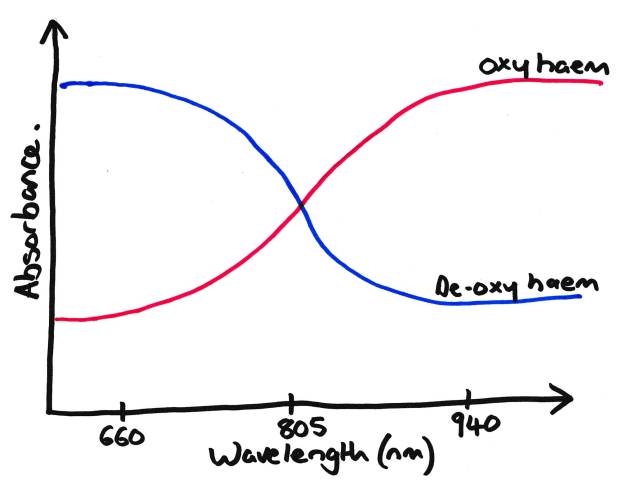
Simplified Diagram showing absorption of both Oxy- and Deoxy-Haemaglobin
You may see some more complicated graphs than the one above in certain books. This one is heavily simplified, but gets the point across in an exam, See the exam technique page for more…
As you can see from the graph above, the absorption of Deoxy-Hb and Oxy-Hb is the same at 805nm, the so called Isobestic point. Analysis of the absorption of infrared at 805nm allows quantification of the total concentration of haemoglobin present. This is how some blood gas machines and continuous non invasive haemoglobin monitors work (if you’ve ever seen one!).
If we look at either end of the graph you can see where Deoxy-Hb has a high absorption and Oxy-Hb a low absorption at 660nm (red), and visa versa at 940nm (infrared). Remember, we’re talking about light in the red to infrared range, 660nm is red. Hence deoxy haemaglobin absorbs lots of red light and appears ‘blue’. Oxy-Hb absorbs little red light (and hence reflects lots) appearing red. That’s a good way to remember which way round they go in the exam.
What you need to know (How it works in practice):
Now all this is fine with a sample of blood but how do we get it to work on your finger? Remember also that we care about the arterial oxygen saturations, not the venous ones, so how do we separate the two? Simple, Remember arterial blood has one defining feature compared to the other parts of your finger…
Its Pulsatile.
So if we graph out everything that will absorb infrared at our spectrum we’re interested in, we see the following:
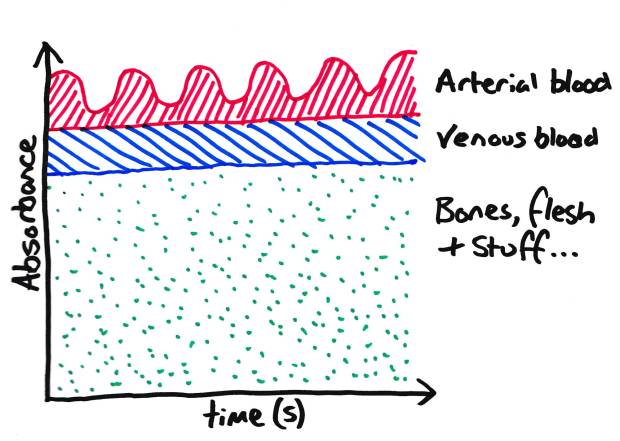
Species which absorb Infrared light in your finger. Note: ‘stuff’ includes nail varnish!
Now, if we discount the non pulsatile absorbents, we are left with arterial blood. This is how the pulse oximeter works, it discounts anything which is not pulsatile.
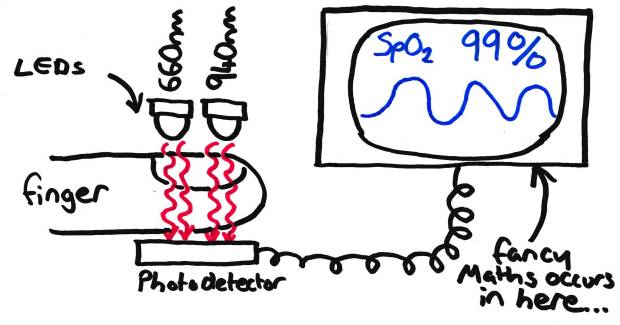
Schematic diagram on Pulse oximeter.
Your typical pulse oximeter will have two LEDS (at 660 and 940nm) which flash alternatively. The photocell/detector on the other side of the finger then sees how much light is absorbed. Most modern devices also cycle 660 > 940 > off to allow compensation for any stray ambient room light that may make its way to the photocell. If you look closely you can see this happening on some models, although most cycle too fast to see easily.
Now if we measure the absorption at each of these wavelengths we can work out (with the aid of some fancy ‘black box’ maths) what the relative concentrations of Oxy and Deoxy-Hb are, and hence, what the oxygen saturations are (as this is simply percentage of haemoglobin that is oxygenated).
The first pulse oximeters were calibrated against blood gas results from medical students who were put in gas chambers with reducing amounts of ambient oxygen. They only allowed their oxygen saturations to drop to the low-mid 80’s (they weren’t that mean!). Below this the results are extrapolated, hence sats probes are notoriously inaccurate below these ranges.
Sources of error with pulse oximeters:
- Pulsatile venous blood flow, think AV fistulas, heart failure, placement of probe substantially lower than heart etc.- Will cause a lower reading due to pulsatile venous (ie deoxygenated) blood.
- Presence of carboxyhaemoglobin will cause over-estimation of saturations (CO-Hb has similar absorption spectra to Oxy-Hb)
- Presence of light leaks (only really in older models which don’t compensate for the presence of ambient light).
- Presence of high bilirubin levels (you may see this on some blood gas analysers in patients with hyperbilirubinaemia!)
- Presence of intravascular dyes (methelene blue and indocyanine green are the classical examples). Both cause artifactually lower readings, usually in the mid 80’s.
- Surgical diathermy!!
- And Many more, There is a very long list.
Random Exam factoids (i.e. the things the college like asking):
- Remember your absorption spectra (660, 805, 940nm). This makes an easy MCQ.
- Remember which way things like carbon monoxide cause sats to go (e.g. overestimate rather than underestimate)
© Sam Beckett and Physics4FRCA, 2017. Unauthorized use and/or duplication of this material without express and written permission from this site’s author and/or owner is strictly prohibited.
