What the curriculum thinks you need to know:
PC_BK_10 Heat: including temperature, absolute zero
PC_BK_11 Heat transfer and loss: conduction, convection, radiation, evaporation
PC_BK_12 Temperature measurement: including Hg, alcohol, infrared, thermistor, thermocouple, Bourdon gauge, liquid crystal. Anatomical sites used for measurement
What you need to know (The theory):
Heat
Type of energy that moves between two objects due to a difference in temperature
Temperature
A measurement of the average kinetic energy of a particles in a system.
OR
Propery of an object which determines if heat energy will flow to or from it from another object
Everyone thinks of temperature in degrees centigrade (of Fahrenheit if you’re from the other side of the pond!). However the SI unit of temperature is the Kelvin.
One kelvin is defined as 1/273.16 of the thermodynamic temperature of the triple point of water (= 0.01oC)
One kelvin is equivalent to one degree centigrade, however the scale starts at different points. zero degrees centigrade is the freezing point of water (0oC), where as zero kelvin is absolute zero (-273.15oC). Which brings us on to….
Absolute Zero
Absolute zero is defined as the temperature at which all molecular movement stops
Absolute zero is a theoretical temperature as you cannot stop ALL molecular motion.
Heat Capacity
The amount of heat energy required to increase the temperature of an object by one Degree Centigrade/Kelvin
Specific Heat Capacity
The amount of heat energy required to increase one Kg of a substance by one Degree Centigrade/Kelvin
Heat transfer and loss
There are numerous ways in which a patient can loose heat during an operation. Remember that patients can loose the ability to produce heat effectively (not completely!) due to muscle relaxation and inability to shiver (although non shivering thermogenesis still occurs). You need to know about each of the methods of heat loss, and how to minimise each of these.
Radiation (~40%)
Transfer of heat due to movement of electromagnetic radiation from the patient.
Convection (~30%)
Heating of the air immediately surrounding the patient. This then rises and allows further air to be heated.
Conduction (~5%)
Due to direct contact and transfer of heat to other surfaces, typically the operating table!
Evaporation (~15%)
Caused by liquids on the body surface (think skin prep or exposed wet bowel) being heated and evaporating. This is why surgeons cover bowel which
Respiratory losses (~10%)
Combination of the above, but from the respiratory tract rather than the body surface.
What you need to know (How it works in practice):
Measurement of temperature
All methods of temperature measurement use properties of certain substances which change with increasing or decreasing temperature.
Mercury/Alcohol
All fluids increase in volume as temperature increases (Think of Charles’ Law from the Gas laws… Volume is proportional to temperature). This is the principle that the simple thermometers we all have seen use. In a tube of liquid, as the temperature increases the liquid expands and moves up the tube. Markers on the tubing signify the height that is attained at a given temperature.
Alcohol is used as it is safer than mercury (less toxic!) but has the downside that the boiling point and freezing point (and hence measurable range) is lower at approx. -115oC to + 78oC.
Mercury on the other hand has a much higher usable range (-27 to 356oC) hence have different uses.
Advantages: Cheap, Don’t require power supply
Disadvantages: Potentially harmful contents, Difficult to position for continuous readout in theatre.
Infrared thermometer
We mentioned earlier that most heat loss is from radiation. Now an infrared detects the proportion of the radiation which is emitted in the infrared spectrum. This is in proportion to the amount of thermal energy present in the item being measured. They have the advantage that they don’t require contact with the patient. They are typically used on the forehead or against the ear drum (the so called ‘tympanic’)
Advantages: Non invasive, Fairly accurate
Disadvantages: Requires power source, Easy to get incorrect readings (usually ear wax is the cause!)
Resistance Wire
The resistance of materials changes with temperature. The easy way to remember this is computers. Computers are cooled to make them work better, they work better as the resistance to current flow decreases with temperatures.
Most resistance thermometers are made out of platinum.
Advantages: Highly accurate (used to calibrate other types)
Disadvantages: Very slow response to change in temperature. Require power supply.

Resistance Wire Thermometer Response Graph
Thermistor
This is a type of semiconductor similar to a transistor, except that the change in temperature changes the resistance to flow of electrical current through it.
Advantages: Fairly accurate, Fast responsive,
Disadvantages: Non linear response. Suffer from drift with time.
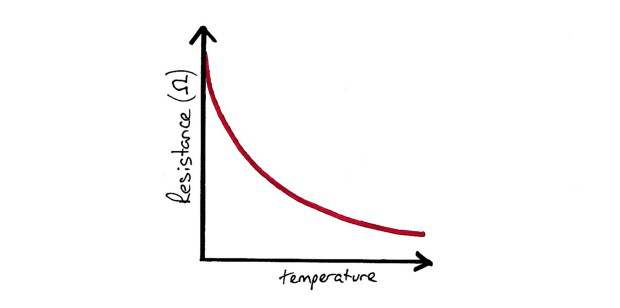
Thermistor Response Graph
Thermocouple
When two dissimilar metals come together, a voltage is produced at the junction. This voltage varies with temperature. This is called the ‘Seebeck Effect’.
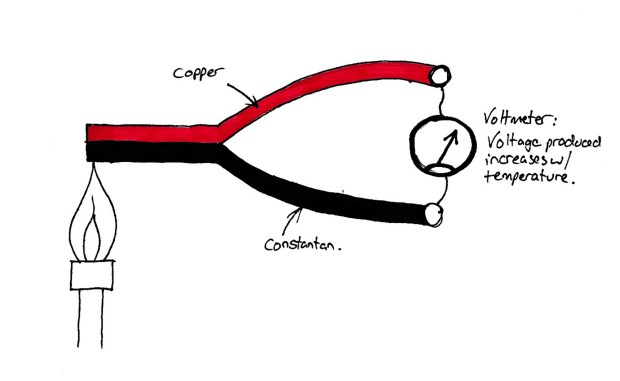
A Simplified Thermocouple
A thermocouple traditionally is made of strips of Constantan (copper/nickel alloy) and copper. The junction of the metals is then placed where the temperature is to be measured. A voltmeter is placed between the opposite ends to measure the potential difference produced. These used to have a second junction (see diagram below) to calibrate it against ambient temperature, however modern thermocouples use an intermal thermistor to calibrate the probe. This is why the typical nasal temperature probes used have two terminals.
Advantages: Cheap, accurate
Disadvantages: Requires power supply (signal needs to be amplified as voltage produced is very small).
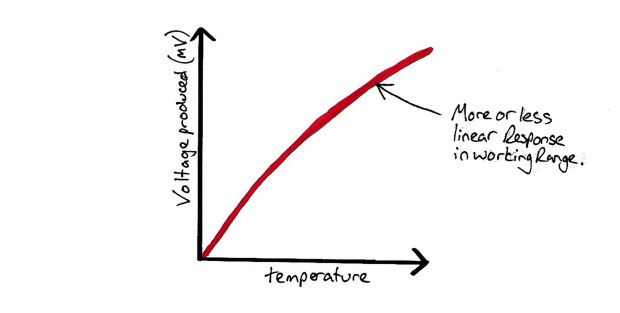
Thermocouple Response Graph
Bourdon ‘gauge’
Now, most people think of pressure measurement with a Bourdon gauge. But, from Charles’ law (see here) we know that the volume of a gas will increase with temperature. So if we close the end of the tube in a bourdon gauge and add a small reservoir of gas to the end (this increases the volume of gas, so increases the volume change with temperature) then we’ve made a thermometer. As the temperture increases, the volume of gas in the tube increases and the tube will try to open, turning the gauge.
Random Exam factoids (i.e. the things the college like asking):
- Definitions, Definitions, Definitions!
- The college like the response graphs to different thermometers and often ask these in VIVAs.
© Sam Beckett and Physics4FRCA, 2017. Unauthorized use and/or duplication of this material without express and written permission from this site’s author and/or owner is strictly prohibited.
